The project enquiry
Creating near-future body implants to change the female body physically and psychologically to reach the goal of creating a ‘perfect female’.
Reflecting on this enquiry, I created three body implants that each address different female stereotypes that society expects females to have. The expectations include females’ rule in household domestic work, females’ pressure on their appearances, and females cannot think independently.
To better explain the functionality of each body implant, I designed User Safety Instructions.
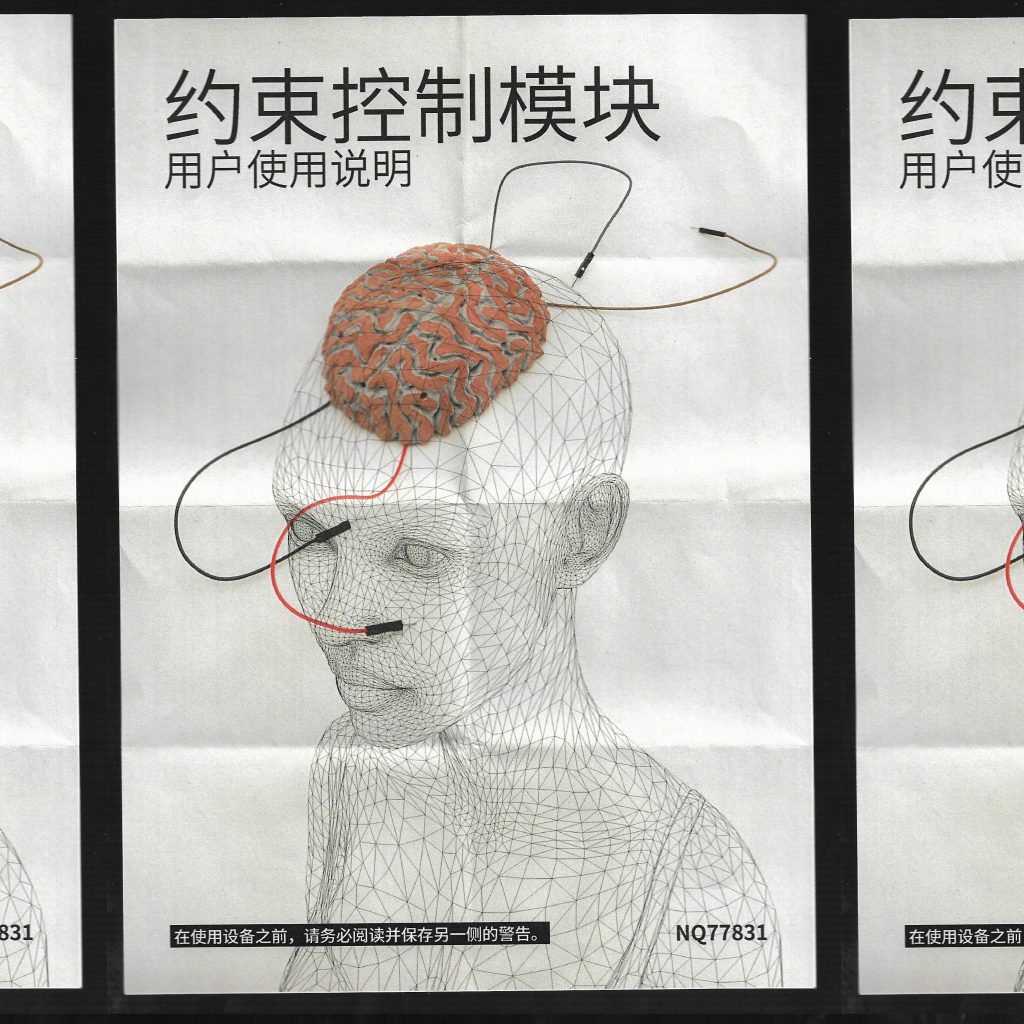

Texts as below:
Bound Control Unit
Please be sure to read and save the instructions prior to using the unit.
Confirm All Accessories
Connection cord × 4
Bound control unit × 1
General use
- The product is a bound control unit for the brain.
- This unit should be connected with the brain to be active.
- This control unit can manipulate the brain and eliminate possible threats when they occur.
- Before use, ensure all components are completely connected.
- This implant can be used by persons with reduced physical, sensory or mental capabilities or lack of experience and knowledge if they have been given supervision or instruction concerning the use of the implant in a safe way and understand the hazards involved.
- Cleaning and user maintenance should not be done by children.
- Do not use the unit for any purpose other than those described in this manual.
- Do not modify or attempt to repair the unit yourself except as indicated in this manual.
- Do not use the unit if it has been modified or damaged. Damaged or modified unit may exhibit unpredictable behaviour resulting in fire, explosion or risk of injury.
Possible Side Effects
Like all implants, this implant can cause side effects, although not everybody gets them.
If the acceptor experiences any hypersensitivity to this implant, i.e., itching, difficulty breathing, swelling of the face, neck, tongue, or throat, or rash (severe allergic reactions), remove it and consult the doctor or pharmacist.
WARNING
Reduce the risk of fire, electric shock, injury, or property damage.
This implant is not considered a lifetime device. The longer acceptors have it, the greater the chances are that they will develop complications, some of which will require more surgery.
Receiving a bound control unit may cause severe brain damage.
This implant has been associated with the development of a cancer of the immune system called brain implant-associated anapaestic large cell lymphoma.
Patients receiving bound control unit implants have reported a variety of systematic symptoms such as joint pain, muscle aches, confusion, chronic fatigue, autoimmune diseases and others. Individual patients’ risk for developing these symptoms has not been well established. Some patients report complete resolution of symptoms when the implants are removed without replacement.
Visit https://gcd.studio/pages/perfect-female or
SCAN HERE USING YOUR SMARTPHONE CAMERA
For full instructions, guarantees and replacement parts.
Bio-Fluid technology warning
May cause allergic reactions to nerves and skin. Wear protective gloves if directly coming into contact with the materials inside the unit. Wash skin thoroughly after handling. Avoid release to the environment. Very is toxic to aquatic life with long-lasting effects, ensure the unit is disposed of correctly as general waste.
MANUFACTURER’S INFORMATION NOTE
Manufacturer’s Conformance Note:
AnKang Bio™ implants are in conformance with Regulation(CN) 2067/963 of the Chinese National Medical Products Administration.
Manufacturer:
AnKang Biotechnology (Group) Co., Ltd.
No.6 Jianguomennei Avenue, Dongcheng District, Beijing, China 100730


Texts as below:
Tactile-aid units for fingers
Please be sure to read and save the instructions prior to using the unit.
Confirm All Accessories
Connection cord × 5
Tactile-aid unit × 5
General use
- The products are tactile-aid units for the fingers.
- This unit can only be active when connected to the hands.
- Before use, ensure all components are completely connected.
- The unit can help improve the appearance of fingers and work as an all-purpose kitchen tool (functions include but are not limited to cutting, chopping, electrifying, firing, temperature and size measuring).
- The unit can force-stop the acceptor’s threatening movements from hurting others and themselves.
- This implant can be used by persons with reduced physical, sensory or mental capabilities or lack of experience and knowledge if they have been given supervision or instruction concerning the use of the implant in a safe way and understand the hazards involved.
- Cleaning and user maintenance should not be done by children.
- Do not use the unit for any purpose other than those described in this manual.
- Do not modify or attempt to repair the unit yourself except as indicated in this manual.
- Do not use the unit if it has been modified or damaged. Damaged or modified units may exhibit unpredictable behaviour resulting in fire, explosion or risk of injury.
Possible Side Effects
Like all implants, this implant can cause side effects, although not everybody gets them.
If the acceptor experiences any hypersensitivity to this implant, i.e., itching, difficulty breathing, swelling of the fingers, hands, forearms, or rash (severe allergic reactions), remove the units and consult the doctor or pharmacist.
WARNING
These implants are not considered lifetime devices. The longer acceptors have it, the greater the chances are that they will develop complications, some of which will require more surgery.
Receiving Tactile-aid units may cause hand disorders, including Tendon disorders, Arthritis, Nerve disorders, and Dupuytren’s disease.
Patients receiving Tactile-aid unit implants have reported a variety of systematic symptoms such as joint pain, muscle aches, confusion, chronic fatigue, autoimmune diseases and others. Individual patients’ risk for developing these symptoms has not been well established. Some patients report complete resolution of symptoms when the implants are removed without replacement.
(the Bio-Fluid technology warning and the Manufacturer information remain the same in all three User Safety Instructions)


Texts as below:
Muscle Tuner Unit
Please be sure to read and save the instructions prior to using the unit.
Confirm All Accessories
Connection cord × 4
Movement sensor chip × 5
Biofiber unit × 1
General use
- The product is a muscle tuner unit for the arms and legs.
- This unit can only be active when connected to the arms or legs.
- Before use, ensure all components are completely connected.
- The unit can tune the movements of muscles.
- The unit can force-stop the acceptor’s threatening movements from hurting others and themselves.
- This implant can be used by persons with reduced physical, sensory or mental capabilities or lack of experience and knowledge if they have been given supervision or instruction concerning the use of the implant in a safe way and understand the hazards involved.
- Cleaning and user maintenance should not be done by children.
- Do not use the unit for any purpose other than those described in this manual.
- Do not modify or attempt to repair the unit yourself except as indicated in this manual.
- Do not use the unit if it has been modified or damaged. Damaged or modified units may exhibit unpredictable behaviour resulting in fire, explosion or risk of injury.
Possible Side Effects
Like all implants, this implant can cause side effects, although not everybody gets them.
If the acceptor experiences any hypersensitivity to this implant, i.e., itching, difficulty breathing, swelling of the arms and legs, body aches and pains, remove the units and consult the doctor or pharmacist.
WARNING
This implant is not considered a lifetime device. The longer acceptors have it, the greater the chances are that they will develop complications, some of which will require more surgery.
Receiving a Muscle Tuner unit may cause body disorders, which include Tendon disorders, Arthritis, Nerve disorders, and critically insufficient blood supply that leads to Gangrene.
Patients receiving Muscle Tuner implants have reported a variety of systematic symptoms such as joint pain, muscle aches, confusion, chronic fatigue, autoimmune diseases and others. Individual patients’ risk for developing these symptoms has not been well established. Some patients report complete resolution of symptoms when the implants are removed without replacement.

